Isotones are the nuclei of atoms of different elements that have the same number of neutrons, but they differ in the mass number.Isobars are the nuclei of atoms of different elements that have the same number of mass ( A ), but they differ in the atomic number ( Z ). 
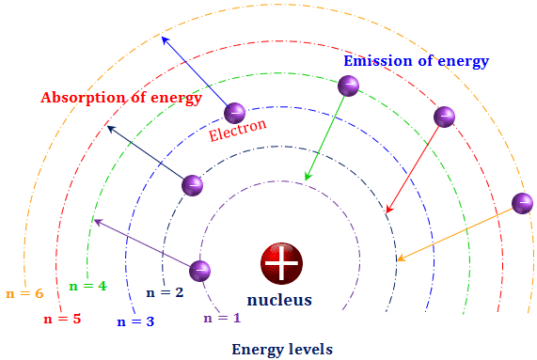
Oxygenelement has three isotopes which are Oxygen-18, Oxygen-16, Oxygen-17, In nuclear chemistry, other nuclear terminology is used in addition to the isotopes, which are : Hydrogen is the simplest element in the nature, It has three isotopes, Name of isotopes are Protium, Deuterium and Tritium, The atomic number equals the mass number in protium nucleus because it does not contain neutrons, Number of neutrons equals the number of protons in deuterium nucleus, while it equals double the number of protons in tritium nucleus. The isotopes have the same chemical properties because they have similar number of electrons and the same electron configuration around the nucleus, Most elements in the periodic table have more than one isotope. Isotopes are the atoms of the same element that have the same atomic number ( Z ), but differ in mass number ( A ) due to the difference in the number of neutrons inside their nuclei. Ītomic number ( Z ) is the number of protons inside the nucleus of the element atom, Number of protons = Number of electrons, Neutrons number is N, N = A – Z, Nucleons are the protons and neutrons inside the nucleus. To describe the nucleus of the atom of any element, you should know Mass number ( A ), Atomic number ( Z ) and Neutrons number ( N ), Mass number ( A ) is the sum of the numbers of protons and nucleus of the element atom. The atom is electrically neutral because the number of positive charged protons in the nucleus equals to the number of negative charged electrons rotating around the nucleus. The atom’s mass is concentrated in the nucleus because the mass of electronsis too small ( negligible ) compared to the mass of protons and neutrons ( mass of protons = 1800 times of electron mass ). Nevil Sidgwick discovered in 1932 that the nucleus contains particles that neutrally charged called neutrons, where the mass of neutron is nearly similar to the mass of proton. Rutherford proved in 1919 that the nucleus of atom contains positively charged particles called protons, where the mass of proton is larger than that of the electronby 1800 times approximately. The negatively charged electrons rotate around the nucleus in certain fixed orbits called energy levels, Each energy level is occupied by a certain number of electronsthat can’t be increased. 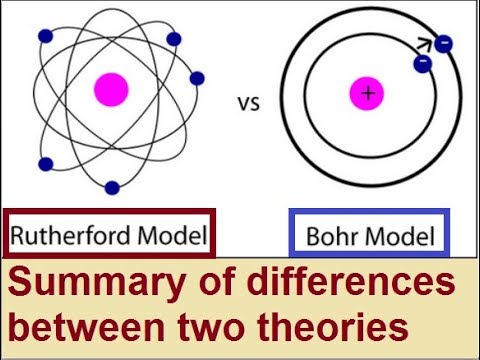
īohr’s Atomic Models Bohr’s atomic models Rutherford’s atomic modelsĪ heavy nucleus carries a positive charge in the centre of atom, Negative electrons rotate around the nucleus at a relatively far distance, Most of the atomic volume is space, The nucleus’s diameter = 10 −6 : 10 −5 nm, while the atom’s diameter = 1 × 10 −10 m ( 0.1 nm ), The mass of the atom is concentrated in the nucleus. Rutherford and Bohr’s Atomic ModelsĪccording to the experiment of Rutherford in 1911 and Bohr’s theory in 1913, the atomic structure became more acceptable. Since the atom is electrically neutral, So, the atom has other particles carrying positive charges equal to the negative charges of the electrons, However, There wasn’t a known distribution for the positive and the negative charges in the atom at that time. Matter is composed of atoms, These atoms show the physical and chemical properties of the matter, By the end of the nineteenth century, Scientists had become sure that electrons are from the main components of atoms, Electrons are negatively charged particles of a very small mass. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
